Cesium Iodide (Metals Basis)
Cesium iodide
CAS: 7789-17-5
Molecular Formula: CsI
Cesium Iodide (Metals Basis) - Names and Identifiers
| Name | Cesium iodide |
| Synonyms | Cesium iodide CESIUM IODINE CESIUM IODIDE caesium iodide tricesiumtriiodide Cesiumiodidewhitextl Cesium iodide, for analysis Cesium Iodide (Metals Basis) |
| CAS | 7789-17-5 |
| EINECS | 232-145-2 |
| InChI | InChI=1/Cs.HI/h;1H/q+1;/p-1 |
Cesium Iodide (Metals Basis) - Physico-chemical Properties
| Molecular Formula | CsI |
| Molar Mass | 259.81 |
| Density | 4.51 g/mL at 25 °C (lit.) |
| Melting Point | 626 °C (lit.) |
| Boling Point | 1280 °C |
| Flash Point | 1280°C |
| Water Solubility | 74 g/100 mL (20 ºC) |
| Solubility | Very soluble in water. Soluble in ethanol. Slightly soluble in methanol. Insoluble in a |
| Vapor Presure | 13.8mmHg at 25°C |
| Appearance | White crystal |
| Specific Gravity | 4.51 |
| Color | White |
| Exposure Limit | ACGIH: TWA 0.01 ppm |
| Merck | 14,2014 |
| PH | pH(50g/l, 25℃) : 5.0~9.0 |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| Stability | Stable. Deliquescent. |
| Sensitive | Hygroscopic |
| Refractive Index | 1.7876 |
| MDL | MFCD00010962 |
| Physical and Chemical Properties | Density 4.51 melting point 626°C boiling point 1280°C water-soluble 74g/100 mL (20°C) |
| Use | Used in the pharmaceutical industry and also as an analytical reagent |
Cesium Iodide (Metals Basis) - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R42/43 - May cause sensitization by inhalation and skin contact. R43 - May cause sensitization by skin contact |
| Safety Description | S22 - Do not breathe dust. S36/37 - Wear suitable protective clothing and gloves. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 3077 9 / PGIII |
| WGK Germany | 2 |
| RTECS | FL0350000 |
| FLUKA BRAND F CODES | 8 |
| TSCA | Yes |
| HS Code | 28276000 |
| Toxicity | LD50 i.p. in rats: 1.4 g/kg (Cochran) |
Cesium Iodide (Metals Basis) - Nature
Open Data Verified Data
cubic crystals or powders. Relative density 4.5 I melting point 621 °c; Boiling point about 1280 °c. Soluble in water, soluble in ethanol, methanol-soluble, almost insoluble in acetone.
Last Update:2024-01-02 23:10:35
Cesium Iodide (Metals Basis) - Preparation Method
Open Data Verified Data
prepared by heating reaction of cesium carbonate and hydroiodic acid, concentrating by evaporation, cooling crystallization and centrifugation.
Last Update:2022-01-01 08:54:12
Cesium Iodide (Metals Basis) - Introduction
Soluble in water and alcohol.
Last Update:2022-10-16 17:28:50
Cesium Iodide (Metals Basis) - Use
Open Data Verified Data
used for infrared spectrometer prism, X-ray fluorescence screen, scintillation counter. For special glass, medicine and used as analytical reagents.
Last Update:2022-01-01 08:54:13
Cesium Iodide (Metals Basis) - Safety
Open Data Verified Data
rats were intraperitoneally injected with LD5o: 1.4g/kg; Rats were orally injected with LD5o:2386mg/kg.
Last Update:2022-01-01 08:54:13
Cesium Iodide (Metals Basis) - Reference Information
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Introduction | cesium iodide for short CsI, including CsI(Tl) inorganic halides are widely used as scintillation media for detectors of high energy particles and ionizing radiation. Vacuum deposited CsI(Tl) films were first used for X-ray imaging inside photoelectric converters. In advanced digital X-ray imaging, CsI(Tl) scintillator films match well with Si-based readout arrays due to high scintillation efficiency for X-ray radiation, high spatial resolution and spectral sensitivity, it is widely used in X-ray imaging. |
| Use | cesium iodide crystals are commonly used as Scintillator Materials for electromagnetic dosimeters in particle physics experiments. Cesium iodide can be used in Fourier transform spectroscopy (FT-IR) as a spectrometer. Compared with the more commonly used potassium bromide spectroscopy, cesium iodide has a wider light transmission range, and the wavelength band can be extended to the far infrared ray. However, there is a problem that the optical cesium iodide crystals are very soft and have no solution, and thus it is difficult to produce a flat polished surface. In addition, the optical crystals of cesium iodide must be stored in a drying vessel to prevent the reaction of water with cesium iodide. The surface of cesium iodide is coated with a layer of germanium to minimize the effect of exposure to moisture in the air during the exchange beam splitter. used in the pharmaceutical industry, also used as analytical reagent |
| solubility in water (g/100ml) | grams dissolved per 100ml of water at different temperatures (℃): 44.1g/0 ℃;58.5g/10 ℃;76.5g/20 ℃;96g/30 ℃;124g/40 ℃; 150g/60 ℃;190g/80 ℃;205g/90 ℃ |
| category | toxic substances |
| toxicity grade | poisoning |
| Acute toxicity | oral-rat LD50: 1400 mg/kg; Intraperitoneal-rat LD50: 1400 mg/kg |
| flammability hazard characteristics | toxic iodide smoke from thermal decomposition |
| storage and transportation characteristics | low temperature ventilation and drying |
| fire extinguishing agent | water, carbon dioxide, foam, dry powder |
Last Update:2024-04-10 22:29:15
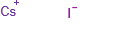
Supplier List
Spot supply
Product Name: Cesium iodide Visit Supplier Webpage Request for quotationCAS: 7789-17-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Cesium iodide Request for quotation
CAS: 7789-17-5
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
CAS: 7789-17-5
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Multiple Specifications
Product Name: Cesium iodide Request for quotationCAS: 7789-17-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Cesium iodide Request for quotation
CAS: 7789-17-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 7789-17-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Spot supply
Product Name: Cesium iodide Visit Supplier Webpage Request for quotationCAS: 7789-17-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Cesium iodide Visit Supplier Webpage Request for quotationCAS: 7789-17-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Cesium iodide Request for quotation
CAS: 7789-17-5
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
CAS: 7789-17-5
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Multiple Specifications
Product Name: Cesium iodide Request for quotationCAS: 7789-17-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Cesium iodide Request for quotation
CAS: 7789-17-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 7789-17-5
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Spot supply
Product Name: Cesium iodide Visit Supplier Webpage Request for quotationCAS: 7789-17-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




